Introduction: Among several intriguing nanoparticles, superparamagnetic iron oxide nanoparticles (SPIONs) have been extensively studied because of their ability to be controlled by magnetic fields. Moreover, SPIONs have also been identified as an excellent contrast agent which can potentially increase image contrast and improve MRI sensitivity[1]. In the current study, an in vitro blood-brain barrier (BBB) model was developed using a triple co-culture of murine brain endothelial cells, astrocytes and pericytes. By measuring the permeability coefficients and TEER (transepithelial electrical resistance) values, we examined the tightness of the cell monolayer and confirmed that this method enables brain endothelial cells to cross-talk with neighboring cells and therefore improve the integrity of the in vitro BBB model. After the model was successfully established and confirmed, we tested several variations of magnetic ferri-liposomes in an effort to increase blood-brain barrier passage for delivering drugs for neural tissue repair while diminishing toxicity.
Materials and Methods: A.Material Characterization: The PEG coated ferri-liposomes were prepared and encapsuled with iron oxide[2]. The nanoparticles were then characterized by zeta potential to determine charge and dynamic light scattering for hydrodynamic diameter. XRD was used to characterize iron oxide magnetite peaks and TEM was used to assess the diameter of both the iron oxide core and liposome. B.Experimental Samples Tested Through the Blood-Brain Barrier Model: For the in vitro blood- brain barrier model, pericytes (R1200, Sciencell) were cultured and seeded on the bottom side of collagen-coated Transwell® inserts using DMEM cell media and astrocytes (CRL-2541, ATCC) were seeded on the bottom of 24-well plates under the same conditions, but separately. After 24 hours of adhesion, murine brain endothelial cells (CRL-2299, ATCC) were seeded onto the upper side of the inserts and the inserts were then placed in the 24-well plates containing astrocytes. The model was evaluated and confirmed using TEER (EVOM2, World Precision Instruments) and FITC-Dextran transport. Then, the model was used to test the permeability of various ferri-liposomes with an iron assay kit (Sigma-Aldrich) used to determine the iron concentration that passed through the model. After the ferri-liposome with the best permeability was chosen, calcein was used as a model drug for drug release tests. Each experiment was conducted in triplicate and repeated at least three times. Differences between means were determined using ANOVA followed by student t-tests.
Results and Discussion: A.Material Characterization: Dynamic light scattering revealed that the hydrodynamic diameters of the liposomes were around 100 nm. XRD indicated the classic magnetite peaks and TEM images showed that the iron core was about 5-10 nm for all of the samples. B.Experimental Samples Tested Through the Blood-Brain Barrier Model: The permeability of FITC-Dextran across the model confirmed that the model was successfully established (Figure 1) and the TEER value of this triple co-cultured model was around 130 Ohms/cm2 (Figure 2). Previous results of samples showed that the highest permeability was obtained from PEG coated nanoparticles. This result suggests that ferri-liposomes coated with PEG should have great permeability across the blood-brain barrier[3]. In addition, strong green fluorescence was associated with the cells incubated with calcein-loaded ferri-liposomes, confirming drug delivery.
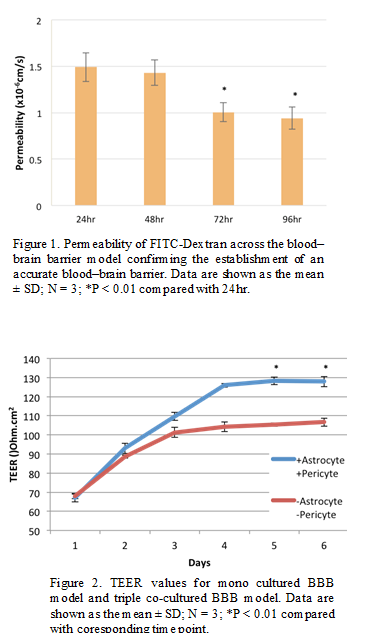
Conclusions: An in vitro model of the blood-brain barrier was established using a triple co-culture method. The model was confirmed by comparing the permeability trend of FITC-dextran in serum-free medium and the TEER value with previous research[4]. The results suggest a possibility to manipulate magnetic nanoparticle penetration across the blood–brain barrier by controlling bioactive coatings. Such data lay the foundation for the modification of ferri-liposome to deliver pharmaceutical agents into the brain.
The authors would like to thank Northeastern University for funding this research.
References:
[1] Richard G, et al. BOLD functional MRI in disease and pharmacological studies: room for improvement?, Magnetic Resonance Imaging, 2007; 25: 978-988.
[2] Georgy Mikhaylov, et al. Ferri-liposomes as an MRI-visible drug-delivery system for targeting tumours and their microenvironment. Nature Nanotechnology 6. 2011; 594–602.
[3] Di S, et al. Controlling ferrofluid permeability across the blood–brain barrier model. Nanotechnology, 2014; 25 (7), 075101.
[4] Bennett J, et al. Blood–brain barrier disruption and enhanced vascular permeability in the multiple sclerosis model EAE. J Neuroimmunol. 2010; 229:180–191.